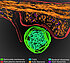
Combating salmonella infections with pathoblockers
Rising antibiotic resistance is making it increasingly difficult to treat salmonella infections. They pose a significant health risk and cost European healthcare systems and the economy billions of euros per year. A team of researchers led by CMFI board member Samuel Wagner has developed a new therapeutic approach that is highly effective and works independently of antibiotic resistance.
The team recently presented their product development, "Anti-virulence agents to prevent and treat Salmonella infections" at BioVaria 2023. BioVaria brings together innovators from academia, leading technology transfer experts, and startups with investors and representatives from the global biotech and pharmaceutical industries.
The team's goal is to bring a novel drug against salmonella infections to market.
Salmonella infections: High numbers of cases are a concern and a problem for the healthcare system and the economy
Salmonella infections are among the most common bacterial diseases in humans, according to the Robert Koch Institute (RKI). In otherwise healthy people, a Salmonella infection usually heals within a few days and without consequences.
In very young and elderly people, however, an intestinal inflammation caused by the infection (salmonellosis) can become a serious problem. If the body's immune system is no longer able to fight the bacterial infection, the diarrhea can lead to life-threatening dehydration. Treatment of the symptoms, such as fever, diarrhea and vomiting, is then no longer sufficient to supply the body with the necessary fluids. In these cases, treatment with antibiotics is indicated.
In 2021, more than 60,000 cases of salmonellosis were reported in the European Union, with a hospitalization rate of 20%. The European Food Safety Authority puts the potential burden on the European economy alone at up to EUR 3 billion per year (source: EFSA Report 2021).
Antibiotic resistance makes treatment increasingly difficult
For several years, an increase in resistance of Salmonella to certain antibiotics has been observed. This is making drug treatment of the bacterial infection increasingly difficult, and long hospital stays and severe courses are on the rise.
To solve this problem, the team around Samuel Wagner has developed so-called pathoblockers that can be used in the treatment of a Salmonella infection - regardless of the resistance of the bacteria to antibiotics.
A new technology: pathoblockers
The pathoblocker developed by the team is a gene-regulating molecule that specifically prevents the formation of the so-called type III secretion system (T3SS). The T3SS is a protein structure on the cell surface that, among other things, has a kind of injection needle by means of which pathogenic bacteria can inject their proteins into host cells. Bacteria such as Salmonella can thus transfer their pathogenic properties into the foreign cell. By preventing the formation of the T3SS, pathoblockers deprive the bacteria of an infectious property.
Pathoblockers combat pathogens efficiently and in a targeted manner. In contrast to antibiotic treatment, the natural intestinal flora is not damaged, and no toxicity of the active ingredient has been detected in laboratory tests to date. In addition, pathoblockers could reduce the likelihood of further antibiotic resistance development, as fewer antibiotics would need to be taken over a shorter period of time. Nevertheless, combination with a standard antibiotic to treat invasive and antibiotic-resistant Salmonella infections is possible.
Conceivably, the drug could be used to prevent traveler's diarrhea caused by Salmonella, for stand-alone treatment, and to shorten a hospital stay for a Salmonella infection.
When will the drug hit the market?
"We have really great and clear study results so far," says Samuel Wagner. "The first very important milestones were demonstrating the efficacy of our compound and elucidating how it works." However, product development is a process that is regulated, and clinical trials could start in 2028 after further optimization of the product, Wagner says. "Assuming investors can be found to develop these new anti-infectives, that could open up a market of 300 million euros annually."
Prof. Samuel Wagner, PhD
University of Tübingen
Interfaculty Institute of Microbiology and Infection Medicine Tübingen
Cellular and Molecular Microbiology
Elfriede-Aulhorn-Str. 6
72076 Tübingen
samuel.wagner@med.uni-tuebingen.de
Website
Leon Kokkoliadis
Public Relations Management
Tel: +49 7071 29-74707
E-Mail: leon.kokkoliadis@uni-tuebingen.de
Related Articles
Springer Nature



Springer Nature